
In April 2018, a research team from Nagasaki University and other institutions in Japan published a breakthrough study in the journal Molecular Neurobiology, titled "Identification of Alprenolol Hydrochloride as an Anti-prion Compound Using Surface Plasmon Resonance Imaging". They used the advanced PlexArray HT system to screen out a potential anti-prion compound from 1,200 FDA-approved drugs - Alprenolol Hydrochloride, a beta-blocker originally used to treat hypertension. This study not only brings new hope for the treatment of prion disease (a fatal neurodegenerative disease), but also demonstrates the great potential of modern technology in drug repositioning.
Research background: Challenges of prion disease
Prion disease is a class of neurodegenerative diseases caused by the aggregation of abnormal prion proteins (PrPSc), including Creutzfeldt-Jakob disease (CJD) in humans, scrapie in sheep, and bovine spongiform encephalopathy (BSE, commonly known as "mad cow disease"). There is currently no effective treatment for these diseases, and patients often deteriorate rapidly after diagnosis. Researchers have been looking for drugs that can inhibit the conversion of PrPc (normal prion protein) to PrPSc, but progress has been slow.
The key role of PlexArray HT: a "detective" for high-throughput screening
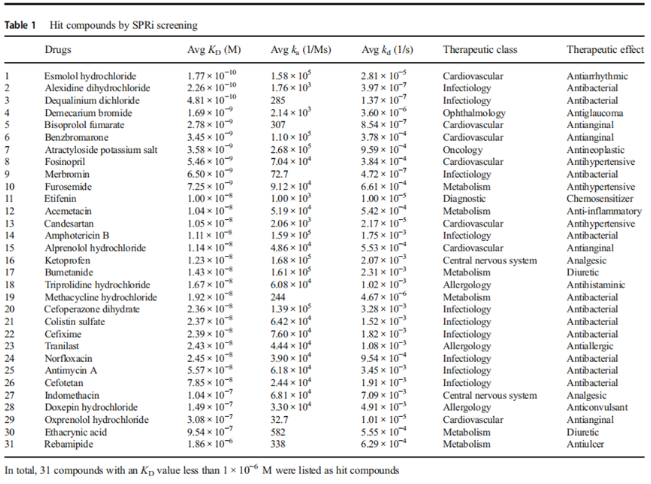
The research team used the PlexArray HT system, a high-throughput screening tool based on surface plasmon resonance imaging (SPRi) technology, to conduct a comprehensive test of 1,200 FDA-approved drugs. SPRi technology is like a sensitive "detective" that can capture binding events between small molecules and proteins in real time. The advantage of PlexArray HT is that it can analyze the binding ability of hundreds of compounds simultaneously in a single experiment, which is much more efficient than traditional SPR technology.
In the experiment, the researchers used recombinant human prion protein (rHuPrP90-231) as a target and a drug library fixed on a sensor chip as a "suspect". Through the scanning of PlexArray HT, they found that 31 drugs showed strong binding activity with PrP, among which alpronolol hydrochloride ranked first with a dissociation constant (KD) of 1.14×10-8 M.
In vitro verification: alpronolol hydrochloride shines
After screening the candidate compounds, the research team tested their anti-prion activity in prion-infected neuroblastoma cells (N2a-FK cells). The results showed that alpronolol hydrochloride, bisoprolol fumarate and colistin sulfate could significantly reduce the accumulation of PrPSc. In particular, alpronolol hydrochloride showed a strong anti-prion effect at a concentration of 15μM with a half inhibitory concentration (IC50). Even more exciting is that after continuous culture, alpronolol hydrochloride can completely eliminate PrPSc in cells.
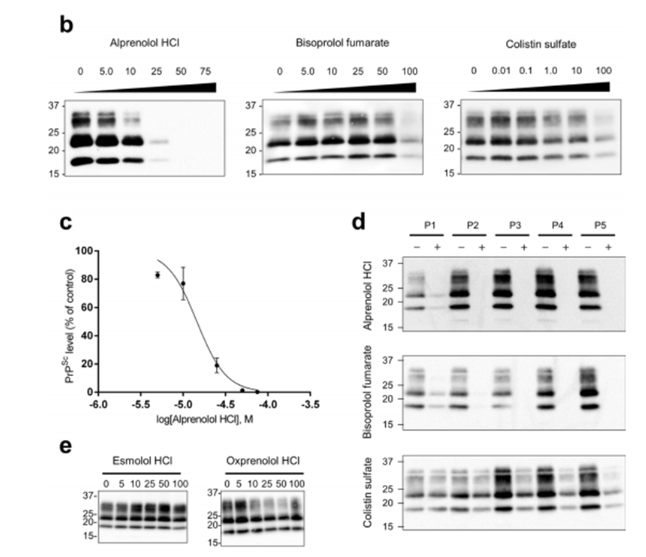
Figure 1b - The inhibitory effect of alpronolol hydrochloride and other compounds on PrPSc accumulation.
Figure 1c - IC50 curve of alpronolol hydrochloride.
Figure 1d - Clearance effect of PrPSc after continuous culture.
In vivo experiment: a ray of hope in the mouse brain
To verify the in vivo effect of alpronolol hydrochloride, the researchers divided CD-1 mice infected with Fukuoka-1 strain prion into three groups: control group (no drug), 50mg/L and 250mg/L alpronolol hydrochloride groups. The drug was administered orally through drinking water, starting from the second day after infection. At 115 days after infection (mid-disease), the researchers found that:
Alpronolol hydrochloride significantly reduced the level of PrPSc in the mouse brain. Immunohistochemical staining showed that PrPSc deposition was significantly reduced in the drug group. Spongiform degeneration (a typical pathological feature of prion disease) was significantly reduced in the cortex of the 50mg/L group.
Nevertheless, alpronolol hydrochloride failed to prolong the survival of mice, which may be related to the reduced water intake in the late stage of the disease.
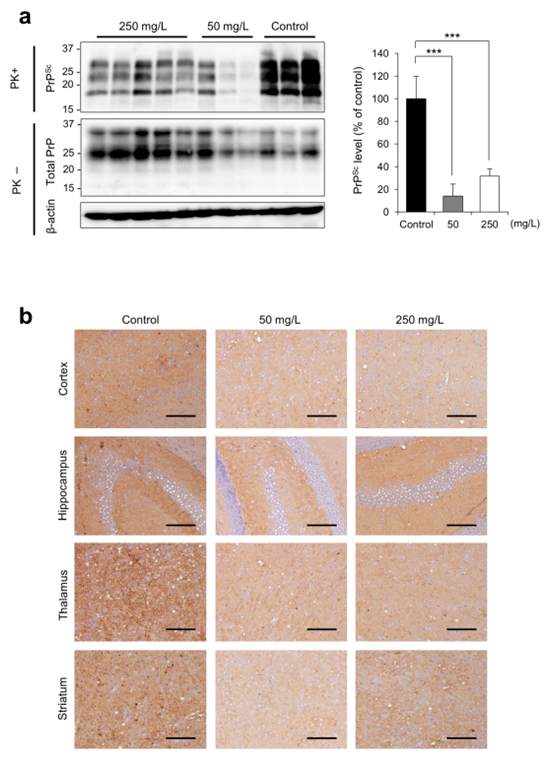
Figure 2a - Western Blot results of PrPSc in mouse brain at 115 days.
Figure 2b - Immunohistochemical staining of PrPSc at 115 days.
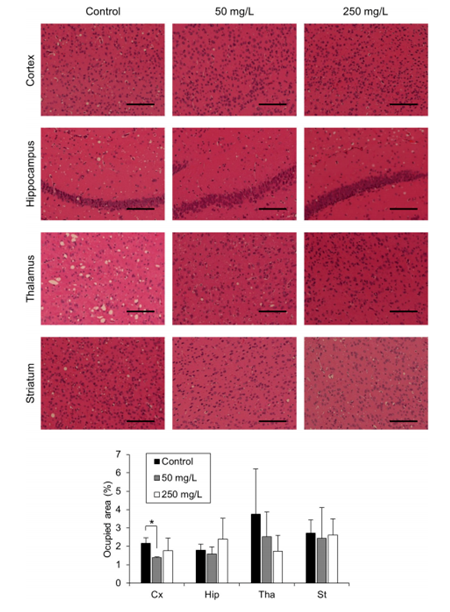
Figure 3 - Quantitative analysis of spongiform degeneration.
Molecular mechanism: docking simulation reveals binding site
Docking simulation analysis performed by AutoDock4.2 showed that alpronolol hydrochloride binds to the "hot spot" region (near helix-B) of mouse PrPc, which is considered to be a key site for PrP conformational transformation. In contrast, the structurally similar oxyprolol (without antiprion activity) interacts only with helix-B with higher binding energy (-5.22 and -4.83 kcal/mol vs. -5.68 and -5.81 kcal/mol for alpronolol hydrochloride), which may explain the difference in their activities.
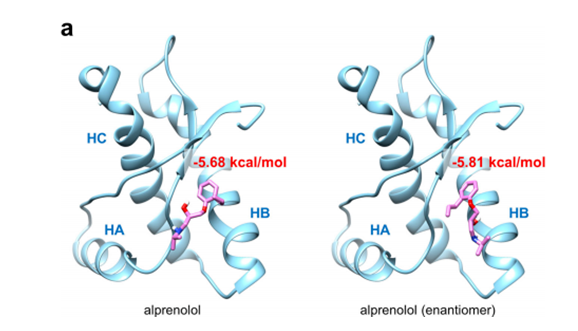
Figure 4a - Docking simulation structure of alpronolol hydrochloride and PrPc.
Summary: PlexArray HT opens up new uses for drugs
This study demonstrates the powerful ability of the PlexArray HT system in drug screening, accurately mining alprenolol hydrochloride as an anti-prion drug candidate from 1,200 existing drugs. It not only cleared PrPSc in vitro, but also showed the potential to inhibit disease progression in mouse models. The research team pointed out that the high-throughput screening capability of PlexArray HT provides new ideas for drug development for neurodegenerative diseases. In the future, further optimization of the dosing regimen may allow alprenolol hydrochloride to be transformed from a blood pressure drug into a "savior" for prion disease.
References Miyazaki, Yukiko et al. "Identification of Alprenolol Hydrochloride as an Anti-prion Compound Using Surface Plasmon Resonance Imaging." Molecular neurobiology vol. 56, 1 (2019): 367-377.
